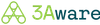Providers
3Aware Clinical Scientific Workbench
3Aware provides a proprietary, AI-driven Clinical Scientific Workbench powered by real-world data from deep, federated health system integrations across a collaborative research network. This approach strengthens investigator-led research within partner institutions while enabling MedTech manufacturers to remotely run in silico, non-interventional observational studies on their products as used at the point of care.
Our real-world evidence generation and analysis solution leverages comprehensive longitudinal patient records - structured and unstructured data - that remain within each health system, and are processed in the health system's own secure cloud environment. With 3Aware's Clinical Scientific Workbench, researchers can rapidly define relevant cohorts, preserve full chronological and episode-of-care context, and use interpretive AI to efficiently surface and intuitively understand the determinant signals and insights embedded across each patient's longitudinal medical record.
Internal Patient Records
Comprehensive Identifiable
Structured & Unstructured Data
Federated Data Alliance Network
Comprehensive & De-Identified
Clinical Scientific Workbench
Study-Specific Patient Cohorts
On-Demand Studies
Identify Relevant Records
AI-Driven Automation
Surface Determinant Content
Natural Language Understanding
Generate Landmark Findings
Intuitive Clinical Analysis
Scientific
ResearchRegulatory
StudiesComparative
EffectivenessValue
Analysis
Clinical Scientific Workbench
From Data Custody to Measurable Impact
Facilitating highly-effective clinical studies to enhance institutional research and fuel MedTech commercialization, the 3Aware Clinical Scientific Workbench creates a secure, repeatable path from raw clinical context to usable evidence.
- 1
Resides and operates within your secure cloud infrastructure, allowing patient data and processing to remain under your control, with strict oversight of exports.
- 2
Ingests comprehensive longitudinal patient records—both structured and unstructured multi-modal content—from your EHRs and clinical/management systems, in your format/ontology.
- 3
Identifies patient-level experiences with specific indications, devices, procedures, etc.; with complete on-demand context of the longitudinal patient journey—enabling highly efficient analysis of end-point specific cohorts.
- 4
Leverages AI interpretation and automation to efficiently surface critical observations relative to specified end-points, and accurately understand indications, utilization, procedures, performance, efficiency and outcomes.
- 5
Presents highly-curated content from the full perioperative timeline, enabling high-fidelity signal detection and accurate recognition of causal relationships.
Federated Collaborative Research Network
A New Clinical Research Infrastructure
The Federated Collaborative Research Network (FCRN), powered by 3Aware, is a clinical research infrastructure where each participating health system can easily and securely:
Not a Data Brokerage.
A Commercial Data Partnership.
Enhance their own clinical research results and productivity.
Facilitate collaborative cross-network research.
Facilitate the responsible commercialization of their patients' data.
Enhance their institutional brand and key opinion leadership.
What does your data strategy look like?
FEDERATED COLLABORATIVE RESEARCH NETWORK
Operational, Research & Strategic Value
Move faster from question to Comparative Effectiveness Research (CER) with repeatable evidence workflows—and generate insights that support value analysis, clinical pathways, and operational decision-making.
Position your institution as a trusted, innovation-forward research entity—publishing high-impact evidence, and strengthening your Key Opinion Leadership (KOL) reputation with patients, clinicians, and industry.
Participate in multi-institution research with an “institutional-first” model—controlled, selective, and governed by each participant.
Enable non-reimbursement revenue through leading MedTech manufacturer-funded research (e.. retrospective subscriptions and prospective studies)—without one-off data sales.
FEDERATED COLLABORATIVE RESEARCH NETWORK
Trust & Security
The FCRN model is built for health system security and governance requirements.
Secure processing in your environment (local data)
Maintain custody of identified and de-identified data
Retain export control over what leaves your environment
SSO compatible and aligned to enterprise access patterns
Role-based access controls (RBAC) and ACLs with least privilege principles
Encryption in transit and at rest
Auditability + export controls to support internal governance and review
Cloud compatible: AWS / Azure / GCP
Become a Commercial Data Partner
Partnering with 3Aware simplifies research, unearths new insights, and unlocks new value—with minimal operational disruption. Ready to learn more?
Let's Talk